12. May 2026 By Dr. Jascha Diemer
From Lab Catalog to Intelligent Assistant: How labRAG Brings GenAI Safely into Diagnostics
If you run a diagnostic lab today, your problem is rarely the lack of content. You already maintain a lab catalog, internal standards, quality documents, payer rules and links to guidelines. The real challenge is that regulation and science move faster than your database: new recommendations, updated cutoffs, additional panels – while your team is busy with routine operations, validation work and the next regulatory audit.
The result is familiar:
- Parts of the “current truth” live in the LIS and orderentry system.
- Other parts are buried in PDFs, slide decks, shared drives and inboxes.
- And a nontrivial share sits “in people’s heads” – senior colleagues filling the gap via phone calls and adhoc consults.
Your referrers can still get from a clinical question to a reasonable order, but it costs time and attention. They scroll through parameter lists, crosscheck external guideline PDFs, and call the lab when things get too fuzzy. Every change in guidelines or reimbursement rules adds one more place that has to be updated manually – and one more opportunity for the catalog to drift away from what you would actually recommend.
With labRAG we address exactly this mismatch between what the lab knows and what the catalog can realistically reflect.
Instead of asking your team to keep a single, “perfect” database in lockstep with every regulatory and scientific update, we add a GenAIbased navigation layer on top of your existing assets. labRAG runs inside the lab portal, takes the clinical question as input and stitches together what is already there: catalog entries, curated guideline snippets, internal standards and contractual constraints. The output is a constrained, sourcebacked proposal for tests and panels – deliberately stopping short of diagnosis or therapy advice.
In other words: we are not replacing your expertise or your catalog. We are giving you a controlled copilot that makes your fragmented, constantly changing knowledge base usable at the point of ordering – without taking the wheel out of your hands.
What we expect from a “diagnostic navigation system”
As someone working in highly regulated fields, you need clarity on what exactly a GenAI component should (and should not) do in your environment.
In our view, a diagnostic navigation system has a very specific job:
- The input is not a street address, but a clinical question: suspected condition, differential diagnosis, followup on an abnormal finding, specific symptom constellation, possibly with comorbidities or medication context.
- The output is not a GPS coordinate, but a defensible set of lab tests or panels that fit this context, your catalog, your internal standards and the applicable guidelines and reimbursement rules.
- The constraints along the way are your reality: professional guidelines, internal SOPs, contractual frameworks, regulatory requirements.
For labRAG, this translated into clear expectations. It should be able to:
- Parse clinical questions in natural language – as they are actually asked by referrers (“unclear anemia in pregnancy”, “followup after elevated ferritin”, etc.).
- Navigate your existing knowledge map – lab catalog, guideline excerpts, internal standards, payer rules – instead of inventing its own medicine.
- Propose one or more “routes” through your catalog – e.g. a set of parameters or panels that make sense for the given suspicion and context, including known interferences (comorbidities, medication) where this is captured in your sources.
Equally important are the boundaries:
- labRAG suggests tests and surfaces background information from your curated sources.
- It does not diagnose, does not interpret individual results as “disease X” and does not recommend therapies or medication.
- Where no safe, wellsupported route exists in the available knowledge, it must be able to abstain and explicitly hand the case back to human expertise.
In short: labRAG is designed as a navigation layer inside your portal – it helps plan the route through your test catalog under your rules. You and your referrers still steer the car and remain fully responsible for where you go.
Why a standard chatbot is not an option for your lab
From a lab management perspective, a “diagnostic assistant” that behaves like a generic chatbot is a liability, not a solution. It’s the equivalent of a navigation system that invents roads, hides its map and is vague about which traffic rules it is following, sometimes you get roughly where you wanted to go, but you would never make it part of a validated process.
Outofthebox large language models are impressive text generators, but they have properties that are hard to accept in a regulated diagnostic environment:
- Hallucinations: The model can produce clinically wrong content – in a tone that sounds more confident than any guideline.
- No transparent sourcing: Standard chatbots typically do not show which sources an answer is based on. Expost verification or audit is almost impossible.
- No regulated scope: Neither functional scope nor risk profile are clearly bounded or documented – exactly what you need for compliance, quality management and EU AI Act readiness.
For labRAG, we turned these weaknesses into explicit design principles:
1. Safety: hard red lines
- No diagnosis, no therapy or medication recommendations.
- No interpretation of individual lab values as “this speaks for disease X”; instead, reference ranges and sources are surfaced.
- Defined behaviour under uncertainty: if a question is outside the covered scope or cannot be safely backed by the connected sources, the system must actively say no and hand the case back to human expertise.
2. Traceability: every answer is reviewable
- Every answer is backed by explicit sources (catalog entries, internal standards, guideline snippets, contracts).
- Users can see what each suggestion is based on – a prerequisite for professional acceptance, QM review and auditability.
3. Extensibility: growing with your lab
- New or updated knowledge sources (e.g. new tests, changed recommendations, new contracts) can be connected without reinventing the system.
- Specialised “route planners” (agents) for particular question types – such as reimbursement, specialised diagnostics or followup scenarios – can be added over time.
- labRAG can evolve with your portfolio, your SOPs and the guideline landscape.
In other words, labRAG is deliberately not “just another chatbot in the portal”, but a controlled navigation layer that fits into your quality and regulatory logic – something you can still defend when an auditor asks how GenAI is used in your lab.
Under the hood: how labRAG works
LabRAG does not improvise; it combines specialized components with a curated medical knowledge base. The system translates clinical questions into structured queries against guidelines, catalogs and provided internal standards, while simultaneously operating systems within clearly define boundaries to not cross into diagnostic or therapy recommendations.
Agents as specialized “route planners”
In a real lab workflow, not every question is the same.
“Which tests should I order?” is a different problem from “What does this parameter measure?” or “What is the ICD10 code here?”. If you want meaningful support, the system has to recognize that difference and route the query accordingly.
labRAG does this with specialized agents – essentially internal “route planners” for different question types:
- Symptom/diagnosis agent: Takes a clinical question (suspicion, differential, followup) and proposes reasonable combinations and sequences of tests, aligned with guideline logic (e.g. AWMF, KBV) and your catalog.
- Lab test content agent: Answers questions about individual parameters or panels: what they measure, typical indications, preanalytical aspects (storage, transport), and how they appear in the catalog.
- ICD10 agent: Supports working with diagnosis codes where they are part of the ordering or documentation workflow.
- Pricing/billing agent: Maps tests to EBM/GOÄ codes and contractual rules, so that ordering stays consistent with billing and reimbursement constraints.
The routing to these agents happens entirely in the background. Users don’t have to pick an “expert”; they ask their question in natural language, and labRAG forwards it to the appropriate internal specialist.
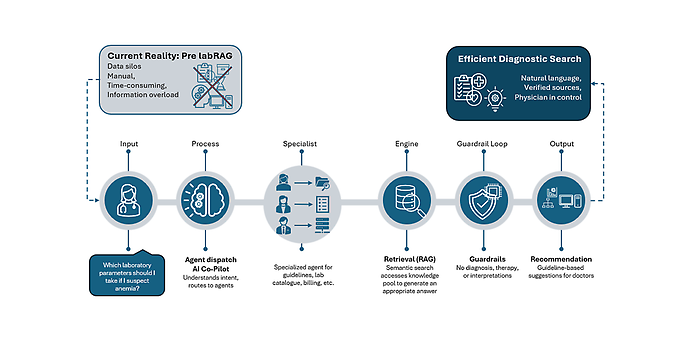
RetrievalAugmented Generation (RAG): consulting the map before answering
A navigation system you would trust does not “guess” the route – it consults the map first. labRAG follows the same principle.
Technically, this is RetrievalAugmented Generation:
- Retrieve: labRAG searches across your curated medical knowledge base (catalog, guideline excerpts, internal standards, contracts) and retrieves the most relevant passages.
- Generate: a language model then uses only these passages to formulate a concise answer.
Because retrieval comes first and generation second, labRAG can always show its sources. Users see not just the final suggestion, but also the underlying documents and standards it is based on.
A dedicated quality check runs in the background: it evaluates whether the retrieved documents actually fit the question. If the match is too weak, labRAG prefers to withhold an answer instead of fabricating a potentially plausible but wrong one.
Flexible, futureproof architecture
Under the hood, labRAG combines:
- 1. a semantic search layer tuned to medical language (synonyms, abbreviations, typical clinical phrasing), and
- 2. a modular agent architecture that allows you to add new capabilities and data sources over time.
The semantic layer helps the system recognize that “autoimmune hepatitis”, “AIH” and a longer clinical description refer to the same diagnostic space, and then retrieve the right parts of your diagnostic “map”. The modular design lets you plug in additional agents (e.g. for new specialties or reimbursement logic) and connect new content sources as your portfolio and guidelines evolve.
A typical interaction might look like this:
- User question: “Suspected autoimmune hepatitis: which tests to use?”
- Internally
- Classification as a clinical question → routed to the symptom/diagnosis agent
- Retrieval of relevant guidelines and catalog entries via RAG
- Answer generation based strictly on these sources
- On screen (for the physician)
- A concise list or table of suggested tests/panels
- Short explanations
- Links to the underlying sources
- Where applicable, hints to relevant billing or coding options
Regulatory compliance
Given its potential impact on diagnostic decisions, labRAG falls into the highrisk category under the EU AI Act and is expected to require conformity assessment by a notified body under the MDR before routine clinical use.
At the time of writing, this classification and the corresponding regulatory pathway are under review with the client. labRAG is not yet approved as a medical device, and the current work focuses on preparing the technical, procedural and documentation basis for a compliant approval process.
If labRAG is used exclusively internally, no conformity assessment needs to be done.
What changes when diagnostics get a navigation layer
From a lab’s perspective, labRAG is less about a “nice AI feature” and more about adding a missing function to the existing infrastructure: navigation.
For your referrers, it means that information which today lives in different places – catalog entries, guideline snippets, internal standards, reimbursement rules – becomes accessible through a single-entry point. A clinical question is entered once and answered with a structured proposal: tests or panels, short explanations, and the relevant constraints and rules. Instead of pushing another standalone pilot, labRAG becomes a pattern for how GenAI can be embedded in diagnostic workflows without undermining control, quality management or regulation.
For you as head of lab, it also opens the door to adjacent, highvalue use cases built on the same principles (domainaware GenAI, explicit sources, hard guardrails):
- GenAI lab training and SOP assistant: A coach that uses your internal SOPs, work instructions and device manuals to generate training content, answer questions on instruments, sample handling or waste disposal, and document completed trainings in a way that stands up in audits.
- Accreditation and qualitymanagement support: An assistant that helps prepare and maintain accreditation and audit documentation by retrieving relevant procedures, validation reports and checklists – effectively a “search + explain” layer on top of your QM system.
- Extended diagnostic and therapy guidance (future scope): Technically, the same architecture could be extended towards guidelinebased diagnostic and therapeutic pathways, combining AWMF recommendations with pharmacological knowledge (e.g. interactions, contraindications) – always within a clearly defined, regulated scope.
All of these follow the same logic as labRAG: a GenAI system that understands your domain, shows its “map”, operates within explicit guardrails and gradually becomes part of everyday workflow – not a detached AI experiment running in parallel to real life in the lab.
Life Sciences
We increase your value creation
We combine strategic consulting, deep industry expertise, and technological implementation – with a focus on measurable value, scalability, and compliance. This is how digital transformation delivers real impact.

